- Tetraoxygen
-
The tetraoxygen molecule (O4), also called oxozone, was first predicted in 1924 by Gilbert N. Lewis, who proposed it as an explanation for the failure of liquid oxygen to obey Curie's law.[1] Today it seems Lewis was off, but not by much: computer simulations indicate that although there are no stable O4 molecules in liquid oxygen, O2 molecules do tend to associate in pairs with antiparallel spins, forming transient O4 units.[2] In 1999, researchers thought that solid oxygen existed in its ε-phase (at pressures above 10 GPa) as O4.[3] However, in 2006, it was shown by X-ray crystallography that this stable phase known as ε oxygen or red oxygen is in fact O8.[4] Nevertheless, tetraoxygen has been detected as a short-lived chemical species in mass spectrometry experiments.[5]
Absorption bands of the O4 molecule e.g. at 360, 477 and 577 nm are frequently used to do aerosol inversions in atmospheric optical absorption spectroscopy. Due to the known distribution of O2 and therefore also O4, O4 slant column densities can be used to retrieve aerosol profiles which can then be used again in radiative transfer models to model light paths.[6]
Free molecule
Theoretical calculations have predicted the existence of metastable O4 molecules with two different shapes: a "puckered" square like cyclobutane,[7] and a "pinwheel" with three oxygen atoms surrounding a central one in a trigonal planar formation similar to boron trifluoride.[8][9] It was previously pointed out that the "pinwheel" O4 molecule should be the natural continuation of the isoelectronic series BO33-, CO32-, NO3-,[10] and analogous to SO3; that observation served as the basis for the mentioned theoretical calculations.
Theoretical structures of metastable O4. 
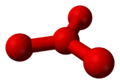
D2d structure D3h structure In 2001, a team at the University of Rome La Sapienza conducted a neutralization-reionization mass spectrometry experiment to investigate the structure of free O4 molecules.[5] Their results did not agree with either of the two proposed molecular structures, but they did agree with a complex between two O2 molecules, one in the ground state and the other in a specific excited state.
References
- ^ Lewis, Gilbert N. (1924). "The Magnetism of Oxygen and the Molecule O2". Journal of the American Chemical Society 46 (9): 2027–2032. doi:10.1021/ja01674a008.
- ^ Oda, Tatsuki; Alfredo Pasquarello (2004). "Noncollinear magnetism in liquid oxygen: A first-principles molecular dynamics study". Physical Review B 70 (134402): 1–19. Bibcode 2004PhRvB..70m4402O. doi:10.1103/PhysRevB.70.134402.
- ^ Gorelli, Federico A.; Lorenzo Ulivi, Mario Santoro, and Roberto Bini (1999). "The ε Phase of Solid Oxygen: Evidence of an O4 Molecule Lattice". Physical Review Letters 83 (20): 4093–4096. Bibcode 1999PhRvL..83.4093G. doi:10.1103/PhysRevLett.83.4093.
- ^ Lars F. Lundegaard, Gunnar Weck, Malcolm I. McMahon, Serge Desgreniers and Paul Loubeyre (2006). "Observation of an O8 molecular lattice in the phase of solid oxygen". Nature 443 (7108): 201–204. doi:10.1038/nature05174. PMID 16971946.
- ^ a b Cacace, Fulvio; Giulia de Petris, and Anna Troiani (2001). "Experimental Detection of Tetraoxygen". Angewandte Chemie International Edition 40 (21): 4062–4065. doi:10.1002/1521-3773(20011105)40:21<4062::AID-ANIE4062>3.0.CO;2-X. PMID 12404493.
- ^ Friess, U. and Monks, P. S. and Remedios, J. J. and Wagner, T. and Platt, U. (2005). "MAX-DOAS O4 measurements: A new technique to derive information on atmospheric aerosols - Retrieval of aerosol properties". Journal of Geosphysical Research.
- ^ Hernández-Lamoneda, R.; A. Ramírez-Solís (2000). "Reactivity and electronic states of O4 along minimum energy paths". Journal of Chemical Physics 113 (10): 4139–4145. doi:10.1063/1.1288370.
- ^ Røeggen, I.; E. Wisløff Nilssen (1989). "Prediction of a metastable D3h form of tetra oxygen". Chemical Physics Letters 157 (5): 409–414. doi:10.1016/0009-2614(89)87272-0.
- ^ Hotokka,M.; P.Pyykkö (May 1989). "Ab initio study of bonding trends in the series BO33-, CO32-, NO3- and O4(D3h)". Chemical Physics Letters 157(5):415-418.
- ^ Jubert,A.H.; E.L.Varetti (1986). "On the possible existence of the O4 molecule with D3h symmetry". Anales de Química (Spain)82:227-230.
See also
Tetraoxygen
O4Octaoxygen
O8Categories:
Wikimedia Foundation. 2010.
