- Natural killer cell
-
Natural killer cell Latin lymphocytus K Code TH H2.00.04.1.02006 Natural killer cells (or NK cells) are a type of cytotoxic lymphocyte that constitute a major component of the innate immune system. NK cells play a major role in the rejection of tumors and cells infected by viruses. They kill cells by releasing small cytoplasmic granules of proteins called perforin and granzyme that cause the target cell to die by apoptosis (programmed cell death).
NK cells are defined as large granular lymphocytes (LGL) and constitute the third kind of cells differentiated from the common lymphoid progenitor generating B and T lymphocytes.[1] They do not express T-cell antigen receptors (TCR) or Pan T marker CD3 or surface immunoglobulins (Ig) B cell receptors but they usually express the surface markers CD16 (FcγRIII) and CD56 in humans, NK1.1 or NK1.2 in C57BL/6 mice. Up to 80% of human NK cells also express CD8.
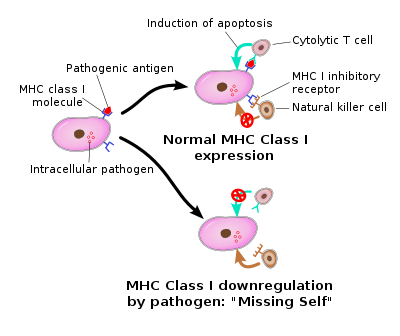
They were named "natural killers" because of the initial notion that they do not require activation in order to kill cells that are missing "self" markers of major histocompatibility complex (MHC) class I.
They are distinct from Natural Killer T cells.
Contents
Activation
Given their strong cytolytic activity and the potential for auto-reactivity, NK cell activity is tightly regulated. NK cells must receive an activating signal, which can come in a variety of forms, the most important of which are listed below.
- The cytokines play a crucial role in NK cell activation. As these are stress molecules released by cells upon viral infection, they serve to signal to the NK cell the presence of viral pathogens. Cytokines involved in NK activation include IL-12, IL-15, IL-18, IL-2, and CCL5.
- NK cells, along with macrophages and several other cell types, express the Fc receptor (FcR) molecule (FC-gamma-RIII = CD16), an activating biochemical receptor that binds the Fc portion of antibodies. This allows NK cells to target cells against which a humoral response has been mobilized and to lyse cells through Antibody-dependent cellular cytotoxicity (ADCC).
- Activating and inhibitory receptors
- Aside from the Fc receptor, NK cells express a variety of receptors that serve either to activate or to suppress their cytolytic activity. These receptors bind to various ligands on target cells, both endogenous and exogenous, and have an important role in regulating the NK cell response.
Mechanism
NK cells are cytotoxic; small granules in their cytoplasm contain proteins such as perforin and proteases known as granzymes. Upon release in close proximity to a cell slated for killing, perforin forms pores in the cell membrane of the target cell, creating an aqueous channel through which the granzymes and associated molecules can enter, inducing either apoptosis or osmotic cell lysis. The distinction between apoptosis and cell lysis is important in immunology: lysing a virus-infected cell would only release the virions, whereas apoptosis leads to destruction of the virus inside.
NK cells are activated in response to interferons or macrophage-derived cytokines. They serve to contain viral infections while the adaptive immune response is generating antigen-specific cytotoxic T cells that can clear the infection. Patients deficient in NK cells prove to be highly susceptible to early phases of herpes virus infection.
In order for NK cells to defend the body against viruses and other pathogens, they require mechanisms that enable the determination of whether a cell is infected or not. The exact mechanisms remain the subject of current investigation, but recognition of an "altered self" state is thought to be involved. To control their cytotoxic activity, NK cells possess two types of surface receptors: activating receptors and inhibitory receptors. Most of these receptors are not unique to NK cells and can be present in some T cell subsets as well.
These inhibitory receptors recognize MHC class I alleles, which could explain why NK cells kill cells possessing low levels of MHC class I molecules. This inhibition is crucial to the role played by NK cells. MHC class I molecules consist of the main mechanism by which cells display viral or tumor antigens to cytotoxic T-cells. A common evolutionary adaption to this seen in both intracellular microbes and tumours is a chronic down-regulation of these MHC I molecules, rendering the cell impervious to T-cell mediated immunity. It is believed that NK cells, in turn, evolved as an evolutionary response to this adaption, as the loss of the MHC would deprive these cells of the inhibitory effect of MHC and render these cells vulnerable to NK cell mediated apoptosis.
Receptor types
NK cell receptor types (with inhibitory as well as some activating members) are differentiated by structure:
- CD94 : NKG2 (heterodimers) — a C-type lectin family receptor, conserved in both rodents and primates and identifies non-classical (also non-polymorphic) MHC I molecules like HLA-E. Expression of HLA-E at the cell surface is dependent on the presence of nonamer peptide epitope derived from the signal sequence of classical MHC class I molecules, which is generated by the sequential action of signal peptide peptidase and the proteasome. Though indirect, this is a way to survey the levels of classical (polymorphic) HLA molecules.
- Ly49 (homodimers) — a relatively ancient, C-type lectin family receptor; are of multigenic presence in mice, while humans have only one pseudogenic Ly49; the receptor for classical (polymorphic) MHC I molecules.
- KIR (Killer-cell immunoglobulin-like receptors) — belong to a multigene family of more recently-evolved Ig-like extracellular domain receptors; are present in non-human primates; and are the main receptors for both classical MHC I (HLA-A, HLA-B, HLA-C) and also non-classical HLA-G in primates. Some KIRs are specific for certain HLA subtypes.
- ILT or LIR (leukocyte inhibitory receptors) — are recently-discovered members of the Ig receptor family.
NK cell receptors can also be differentiated based on function. Natural cytotoxicity receptors directly induce apoptosis after binding to ligands that directly indicate infection of a cell. The MHC dependent receptors (described above) use an alternate pathway to induce apoptosis in infected cells.
History and discovery
In early experiments on cell-mediated cytotoxicity against tumor target cells, both in cancer patients and animal models, investigators consistently observed what was termed a "natural" reactivity, that is, a certain population of cells seemed to be able to lyse tumor cells without having been previously sensitized to them. As these discoveries were incompatible with the established model at the time, many initially considered that these observations were artifacts.[2] However, by 1973, 'natural killing' activity was established across a wide variety of species, and the existence of a separate lineage of cells possessing this ability was postulated.
The discovery that a unique type of lymphocyte was responsible for “natural” or spontaneous cytotoxicity was made in the early 1970s by doctoral student Rolf Kiessling and post-doctoral fellow Hugh Pross, in the mouse,[3] and by Hugh Pross and doctoral student Mikael Jondal in the human. [4][5] The mouse and human work was carried out under the supervision of professors Eva Klein and Hans Wigzell, respectively, of the Karolinska Institute, Stockholm. Kiessling’s research involved the well-characterized ability of T-lymphocytes to lyse tumor cells against which they had been previously immunized. Pross and Jondal were studying cell-mediated cytotoxicity in normal human blood and the effect of the removal of various receptor-bearing cells on this cytotoxicity. Later that same year Ronald Herberman published similar data with respect to the unique nature of the mouse effector cell. [6] The human data were confirmed, for the most part, by West et al.[7] using similar techniques and the same erythroleukemic target cell line, K562. K562 is highly sensitive to lysis by human NK cells and, over the decades, the K562 51Chromium-release assay has become the most commonly used assay to detect human NK functional activity.[8] Its almost universal use has meant that experimental data can be compared easily by different laboratories around the world.
Using discontinuous density centrifugation and, later, monoclonal antibodies, natural killing ability was mapped to the subset of large, granular lymphocytes known today as NK cells. The demonstration that density gradient-isolated large granular lymphocytes were responsible for human NK activity, made by Timonen and Saksela in 1980,[9] was the first time that NK cells had been visualized microscopically and was a major breakthrough in the field.
The cells were named "natural killer" because of the initial notion that they do not require activation in order to kill cells that are "missing-self" recognition ("missing-self" recognition is a term used to describe cells with low levels of MHC class I cell surface marker molecules — a situation that could arise due to viral infection, or in tumors under strong selection pressure of killer T cells).
With the discovery of activating receptors almost two decades after the discovery of the inhibitory receptors these cells continue to be called by the same name, though “natural” no longer means the same thing. The term “natural killer” continues to be justified by:
- a morphology characteristic of activated cytotoxic lymphocytes, e.g., large size, high protein synthesis activity in the abundant endoplasmic reticulum (ER), and preformed granules
- the mature state (does not require much new protein synthesis and remodeling before starting to kill)
- the rapid killing activity observed in freshly-isolated NK cells.
See also
- Active Hexose Correlated Compound
- Granzymes
- Hematopoiesis
- Immune system
- Immunoglobulin, e.g., Ig (above)
- Interleukin, e.g., ILx (above)
- Lymphatic System
Literature
- Cellular and Molecular Immunology by Abbul K. Abbas & Andrew Lichtman Saunders Copyright 2003
- How the Immune System Works, 2nd edition, by Lauren Sompayrac, PhD Blackwell Publishing 2003
- Immunobiology: The Immune System In Health And Disease by Janeway, Travers, Walport & Shlomchik Churchchill Livingstone Copyright 2005
- Kuby Immunology, 6th edition, by Thomas J. Kindt, Richard A. Goldsby,and Barbara A.Kuby W.H. Freeman and Company,New York
- Regulation of interferon-gamma during innate and adaptive immune response, by Schoenborn J.R., and Wilson C.B. Advances in Immunology, 96:41-101, 2007. PMID 7981204
References
- ^ Roitt I, Brostoff J, Male D (2001). Immunology (6th ed.), 480p. St. Louis: Mosby, ISBN 0723431892.
- ^ Oldham R (1983). "Natural killer cells: Artifact to reality: An odyssey in biology". Cancer Metastasis Reviews 2 (4): 323–36. doi:10.1007/BF00048565. PMID 6375859.
- ^ Kiessling, R., Klein, E., Pross, H. and Wigzell, H. "Natural" killer cells in the mouse. II. Cytotoxic cells with specificity for mouse Moloney leukemia cells. Characteristics of the killer cell. Eur. J. Immunol. 5:117-121, 1975. PMID 1086218
- ^ Pross, H.F., and Jondal, M. Cytotoxic lymphocytes from normal donors. A functional marker of human non-T lymphocytes. Clin. exp. Immunol. 21:226- 235, 1975. PMID 810282
- ^ Jondal, M. and Pross, H. Surface markers on human B and T lymphocytes. VI. Cytotoxicity against cell lines as a functional marker for lymphocyte subpopulations. Int. J. Cancer 15:596-605, 1975. PMID 806545
- ^ Herberman, R.B., Nunn, M.E., Holden, H.T., and Lavrin, D.H. Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic and allogeneic tumors. II. Characterization of effector cells. Int. J. Cancer 16:230-239, 1975. PMID 1080480
- ^ West W.H., Cannon, G.B., Kay, H.D., Bonnard, G.D., and Herberman R.B. Natural cytotoxic reactivity of human lymphocytes against a myeloid cell line: characterization of effector cells. J. Immunol. 118:355-61, 1977. PMID 29976
- ^ Pross, H.F., Baines, M.G., Rubin, P., Shragge, P., and Patterson, M. Spontaneous human lymphocyte-mediated cytotoxicity against tumor target cells. IX. Quantitation of natural killer (NK) cell activity. J. Clin. Immunol. 1:51-63, 1981. PMID 7334070
- ^ Timonen, T., and Saksela, E. Isolation of human NK cells by density gradient centrifugation. J. Immunol. Methods 36: 285-291, 1980. PMID 7430655
External links
- CopeWithCytoKines Portal to definitions of NK-Cells and closely related topics
- http://www.hfea.gov.uk/fertility-treatment-options-reproductive-immunology.html Reproductive immunology and fertility treatment
- http://www.cambridgenetwork.co.uk/news/article/default.aspx?objid=58465
- Binns C (June 19, 2006), Natural Body Guards: How Your Killer Cells Get Motivated. Livescience.com. Retrieved on 2007-10-20.
- MeSH Natural+Killer+Cells
- Nkcells.info - MediaWiki based information platform specializing on natural killer cells
- Large granular lymphocyte entry in the public domain NCI Dictionary of Cancer Terms
Blood: Lymphocytes Lymphoid/
HSC:CFU-LNK cellsLymphopoiesis Immunology: Lymphocytic adaptive immune system and complement Lymphoid AntigensAntibodiesImmunity vs.
toleranceaction: Immunity · Autoimmunity · Alloimmunity · Allergy · Hypersensitivity · Inflammation · Cross-reactivity
inaction: Tolerance (Central, Peripheral, Clonal anergy, Clonal deletion, Tolerance in pregnancy) · ImmunodeficiencyLymphocytes Substances Complement Categories:- Lymphocytes
- Human cells
Wikimedia Foundation. 2010.