- Dihydroxyacetone
-
Dihydroxyacetone 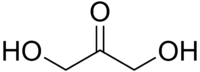 1,3-Dihydroxypropan-2-oneOther namesDihydroxyacetone
1,3-Dihydroxypropan-2-oneOther namesDihydroxyacetone
DHA
GlyceroneIdentifiers CAS number 96-26-4 
PubChem 670 ChemSpider 650 
UNII O10DDW6JOO 
EC-number 202-494-5 DrugBank DB01775 KEGG D07841 
ChEBI CHEBI:16016 
ChEMBL CHEMBL1229937 
Jmol-3D images Image 1 - O=C(CO)CO
Properties[1] Molecular formula C3H6O3 Molar mass 90.078 g/mol Melting point 89-91 °C, 362-364 K, 192-196 °F
Hazards[2] GHS pictograms 
GHS signal word WARNING GHS hazard statements H319 GHS precautionary statements P264, P280, P305+351+338, P337+313  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dihydroxyacetone
 i/ˌdaɪhaɪˌdrɒksiˈæsɨtoʊn/, or DHA, also known as glycerone, is a simple carbohydrate (a triose) with formula C3H6O3.
i/ˌdaɪhaɪˌdrɒksiˈæsɨtoʊn/, or DHA, also known as glycerone, is a simple carbohydrate (a triose) with formula C3H6O3.DHA is primarily used as an ingredient in sunless tanning products. It is often derived from plant sources such as sugar beets and sugar cane, and by the fermentation of glycerin.
Contents
Chemistry
DHA is a hygroscopic white crystalline powder. It has a sweet cooling taste and a characteristic odor. It is the simplest of all ketoses and, having no chiral center, is the only one that has no optical activity. The normal form is a dimer which is slowly soluble in one part water and 15 parts ethanol.[3] When freshly prepared, it reverts rapidly to the monomer in solution. The monomer is very soluble in water, ethanol, diethyl ether and acetone and toluene.
DHA may be prepared, along with glyceraldehyde, by the mild oxidation of glycerol, for example with hydrogen peroxide and a ferrous salt as catalyst. It can also be prepared in high yield and selectivity at room temperature from glycerol using a cationic neocuproine-based palladium catalyst with oxygen or air acting as the co-oxidant.[4] Glyceraldehyde is a structural isomer of dihydroxyacetone.
Biology
Its phosphate form, dihydroxyacetone phosphate (DHAP) takes part in glycolysis, and it is an intermediate product of fructose metabolism.
Uses
DHA was first recognized as a skin coloring agent by German scientists in the 1920s. Through its use in the X-ray process, it was noted as causing the skin surface to turn brown when spilled.
In the 1950s, Eva Wittgenstein at the University of Cincinnati did further research with dihydroxyacetone.[5][6][7][8] Her studies involved using DHA as an oral drug for assisting children with glycogen storage disease. The children received large doses of DHA by mouth, and sometimes spat or spilled the substance onto their skin. Healthcare workers noticed that the skin turned brown after a few hours of DHA exposure.
Eva Wittgenstein continued to experiment with DHA, painting liquid solutions of it onto her own skin. She was able to consistently reproduce the pigmentation effect, and noted that DHA did not penetrate beyond the stratum corneum, or dead skin surface layer. Research then continued on DHA's skin coloring effect in relation to treatment for patients suffering from vitiligo.
This skin browning effect is non-toxic, and similar to the Maillard reaction. DHA reacts chemically with the amino acids in the skin, which are part of the protein containing keratin layer on the skin surface. Various amino acids react differently to DHA, producing different tones of coloration from yellow to brown. The resulting pigments are called melanoidins. These are similar in coloration to melanin, the natural substance in the deeper skin layers which brown or "tan", from exposure to UV rays.
Winemaking
Both acetic acid bacteria A. aceti and G. oxydans use glycerol as a carbon source to form dihydroxyacetone. DHA is formed by ketogenesis of glycerol.[9] It can affect the sensory quality of the wine with sweet/etherish properties. DHA can also react with proline to produce a "crust-like" aroma.[10][9][11] Dihydroxyacetone can affect the anti-microbial activity in wine, as it has the ability to bind SO2.[12]
Sunless tanning
Coppertone introduced the first consumer sunless tanning lotion into the marketplace in the 1960s. This product was called “Quick Tan” or “QT”. It was sold as an overnight tanning agent, and other companies followed with similar products. Consumers soon tired of this product due to unattractive results such as orange palms, streaking and poor coloration. Because of the QT experience, many people today still associate sunless tanning with fake-looking orange tans.
In the 1970s the United States Food and Drug Administration (FDA) added DHA permanently to their list of approved cosmetic ingredients.[13]
By the 1980s, new sunless tanning formulations appeared on the market and refinements in the DHA manufacturing process created products that produced a more natural looking color and better fading. Consumer concerns surrounding damage associated with UV tanning options spurred further popularity of sunless tanning products as an alternative to UV tanning. Dozens of brands appeared on drugstore shelves, in numerous formulations.
Today, DHA is the main active ingredient in all sunless tanning skincare preparations. It may be used alone or combined with other tanning components such as erythrulose. DHA is considered the most effective sun-free tanning additive.
Sunless tanning products contain DHA in concentrations ranging from 1% to 15%. Most drugstore products range from 3% to 5%, with professional products ranging from 5% to 15%. The percentages correspond with the product coloration levels from light to dark. Lighter products are more beginner-friendly, but may require multiple coats to produce the desired color depth. Darker products produce a dark tan in one coat, but are also more prone to streaking, unevenness, or off-color tones. The artificial tan takes 2 to 4 hours to begin appearing on the skin surface, and will continue to darken for 24 to 72 hours, depending on formulation type.
Once the darkening effect has occurred, the tan will not sweat off or wash away with soap or water. It will fade gradually over 3 to 10 days, in conjunction with the skin's normal exfoliation process. Exfoliation, prolonged water submersion, or heavy sweating can lighten the tan, as these all contribute to rapid dead skin cell exfoliation (the dead skin cells are the tinted portion of the sunless tan.)
Current sunless tanners are formulated into sprays, lotions, gels, mousses, and cosmetic wipes. Professional applied products include spray tanning booths, airbrush tan applications, and hand applied lotions, gels, mousses and wipes.
DHA does not damage the skin, and is considered a safe skin coloring agent and nutritional supplement. Contact dermatitis is rarely reported,[citation needed] however a recent study showed that DHA causes severe contact dermatitis in Mexican hairless dogs.[14] Most cases of sensitivity are due to other ingredients in the skin product preparation, such as preservatives, plant extracts, dyes or fragrances.[citation needed]
DHA has been approved for cosmetic use by the FDA, the Canadian Health Ministry, and most of the EU member nations.[citation needed]
DHA-based sunless tanning has been recommended by the Skin Cancer Foundation, American Academy of Dermatology Association, Canadian Dermatology Association and the American Medical Association.
DHA safety considerations
For the 24 hours after self-tanner (containing high DHA levels, ~5%) is applied, the skin is especially susceptible to free-radical damage from sunlight, according to a 2007 study led by Katinka Jung of the Gematria Test Lab in Berlin.[15] Forty minutes after the researchers treated skin samples with high levels of DHA they found that more than 180 percent additional free radicals formed during sun exposure compared with untreated skin. Another self-tanner ingredient, erythulose, produced a similar response at high levels. For a day after self-tanner application, excessive sun exposure should be avoided and sunscreen should be worn outdoors, they say; an antioxidant cream could also minimize free radical production. Although some self-tanners contain sunscreen, its effect will not last long after application, and a fake tan itself will not protect the skin from UV exposure.
The study by Jung et al. further confirms earlier results demonstrating that dihydroxyacetone in combination with dimethylisosorbide enhances the process of (sun-based) tanning. This earlier study also found that dihydroxyacetone also has an effect on the amino acids and nucleic acids which is bad for the skin.[16] The free radicals are due to the generation of AGE (Advanced Glycation Products) such as amadori products (a type of AGE) as a result of the reaction of DHA with the skin. AGEs are behind the damage to the skin that occurs with high blood sugar in diabetes where similar glycation occurs.
References
- ^ Weast, Robert C., ed (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-74. ISBN 0-8493-0462-8..
- ^ HSNO Chemical Classification Information Database (New Zealand Environmental Risk Management Authority), http://www.ermanz.govt.nz/Chemicals/ChemicalDisplay.aspx?SubstanceID=11687, retrieved 2009-09-03.
- ^ Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123, 3225
- ^ Painter, R. M., Pearson, D. M. and Waymouth, R. M. (2010), Selective Catalytic Oxidation of Glycerol to Dihydroxyacetone. Angewandte Chemie International Edition, 49: 9456–9459. doi: 10.1002/anie.201004063.
- ^ Eva Wittgenstein and DHA
- ^ WITTGENSTEIN E, GUEST GM (1961). "Biochemical effects of dihydroxyacetone". J. Invest. Dermatol. 37 (5): 421–6. doi:10.1038/jid.1961.137. PMID 14007781.
- ^ GOLDMAN L, WITTGENSTEIN E, BLANEY D, GOLDMAN J, SAWYER F (1961). "Studies of some physical properties of the dihydroxyacetone color complex". J. Invest. Dermatol. 36: 233–4. PMID 13706567.
- ^ WITTGENSTEIN E, BERRY HK (1960). "Staining of skin with dihydroxyacetone". Science 132 (3431): 894–5. doi:10.1126/science.132.3431.894. PMID 13845496.
- ^ a b Drysdale, G.S. and Fleet, G.H. (1988). "Acetic acid bacteria in winemaking: a review". American Journal of Enology Viticulture 39: 143–154.
- ^ Margalith, Pinhas Flavor microbiology Thomas, 1981. ISBN 0-398-04083-4
- ^ Roger B. Boulton, Vernon L. Singleton, Linda F. Bisson, Ralph E. Kunkee Principles and Practices of Winemaking Springer, 1999. ISBN 0-8342-1270-6
- ^ Eschenbruch R, Dittrich HH. (1986). "Metabolism of acetic acid bacteria in relation to their importance to wine quality". Zentrablatt für Mikrobiologie 141: 279–289.
- ^ Code of Federal Regulations, Title 21, Volume 1, §73.1150 Listing of color additives exempt from certification
- ^ Kimura T. "Contact dermatitis caused by sunless tanning treatment with dihydroxyacetone in hairless descendants of Mexican hairless dogs." Environ Toxicol. 2008 Nov 17. [1]
- ^ K Jung, M Seifert, Th Herrling, J Fuchs "UV-generated free radicals (FR) in skin: Their prevention by sunscreens and their induction by self-tanning agents." Spectrochim Acta A Mol Biomol Spectrosc. 2008 May;69(5):1423-8. Epub 2007 Oct 10. [2]
- ^ Benamar N, Laplante AF, Lahjomri F, Leblanc RM (Oct 2004). "Modulated photoacoustic spectroscopy study of an artificial tanning on human skin induced by dihydroxyacetone". Physiological Measurement 25 (5): 1199–210. doi:10.1088/0967-3334/25/5/010. PMID 15535185.
External links
- How Stuff Works
- US FDA/CFSAN - Tanning Pills
- The Skin Cancer Foundation on Sunless tanners
- American Academy of Dermatology on Self Tanners
- DHA and Vitiligo
- DHA as a Vitiligo option
- Self Tanning lotions as a skin safe alternative to UV tanning
- The American Cancer Society on Sunless Tanners
- New Zealand Dermatological Society recommends sunless tanners
Types of carbohydrates General: Geometry Monosaccharides Aldodiose (Glycolaldehyde)Ketotriose (Dihydroxyacetone) · Aldotriose (Glyceraldehyde)Ketopentose (Ribulose, Xylulose)
Aldopentose (Ribose, Arabinose, Xylose, Lyxose)
Deoxy sugar (Deoxyribose)Ketoheptose (Sedoheptulose, Mannoheptulose)>7Multiple Other oligosaccharidesGlucose/Glucan: Glycogen · Starch (Amylose, Amylopectin) · Cellulose · Dextrin/Dextran · Beta-glucan (Zymosan, Lentinan, Sizofiran) · Maltodextrin
Fructose/Fructan: Inulin · Levan beta 2→6
N-Acetylglucosamine: Chitinbiochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/iCategories:- Cosmetics chemicals
- Diols
- Ketoses
- Trioses
Wikimedia Foundation. 2010.
