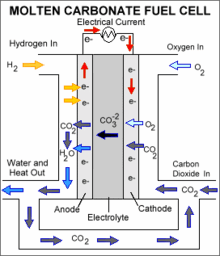
- Molten carbonate fuel cell
-
Molten-carbonate fuel cells (MCFCs) are high-temperature fuel cells, that operate at temperatures of 600°C and above.
Molten carbonate fuel cells (MCFCs) are currently being developed for natural gas, biogas (produced as a result of anaerobic digestion or biomass gasification) , and coal-based power plants for electrical utility, industrial, and military applications. MCFCs are high-temperature fuel cells that use an electrolyte composed of a molten carbonate salt mixture suspended in a porous, chemically inert ceramic matrix of beta-alumina solid electrolyte (BASE). Since they operate at extremely high temperatures of 650°C (roughly 1,200°F) and above, non-precious metals can be used as catalysts at the anode and cathode, reducing costs.
Improved efficiency is another reason MCFCs offer significant cost reductions over phosphoric acid fuel cells (PAFCs). Molten carbonate fuel cells can reach efficiencies approaching 60 percent, considerably higher than the 37-42 percent efficiencies of a phosphoric acid fuel cell plant. When the waste heat is captured and used, overall fuel efficiencies can be as high as 85 percent.
Unlike alkaline, phosphoric acid, and polymer electrolyte membrane fuel cells, MCFCs don't require an external reformer to convert more energy-dense fuels to hydrogen. Due to the high temperatures at which MCFCs operate, these fuels are converted to hydrogen within the fuel cell itself by a process called internal reforming, which also reduces cost.
Molten carbonate fuel cells are not prone to poisoning by carbon monoxide or carbon dioxide —they can even use carbon oxides as fuel— making them more attractive for fueling with gases made from coal. Because they are more resistant to impurities than other fuel cell types, scientists believe that they could even be capable of internal reforming of coal, assuming they can be made resistant to impurities such as sulfur and particulates that result from converting coal, a dirtier fossil fuel source than many others, into hydrogen.
The primary disadvantage of current MCFC technology is durability. The high temperatures at which these cells operate and the corrosive electrolyte used accelerate component breakdown and corrosion, decreasing cell life. Scientists are currently exploring corrosion-resistant materials for components as well as fuel cell designs that increase cell life without decreasing performance.
Contents
MTU fuel cell
The German company MTU Friedrichshafen presented an MCFC at the Hannover Fair in 2006. The unit weighs 20 tons and can produce 240 kW electric power out of different gaseous fuels, biogas included. If fueled by carbon-containing fuels such as natural gas, the exhaust will contain CO2 but reduced by up to 50% compared to diesel engines running on marine bunker fuel.[1] The exhaust temperature is 400 degrees Celsius, enough to be used for many industrial processes. Another possibility is to make more electric power via a Steam turbine. Depending on feed gas type, the electric efficiency is between 42 and 49%. A steam turbine can increase the efficiency up to 64%. The unit can be used for cogeneration.
See also
References
Source
External links
- LLNL: The Carbon/Air Fuel Cell Conversion of Coal-Derived Carbons
- DoD
- MTU 240kW fuel cell presented on the Hannover Fair 2006
- Logan Energy Limited integrate, install and operate all fuel cell technologies
- [1] molten carbonate fuel cells distributed generation challenge
Fuel cells Types Alkaline fuel cell · Blue energy · Direct borohydride fuel cell · Direct carbon fuel cell · Direct-ethanol fuel cell · Direct methanol fuel cell · Electro-galvanic fuel cell · Enzymatic biofuel cell · Formic acid fuel cell · Flow battery · Molten carbonate fuel cell · Microbial fuel cell · Metal hydride fuel cell · Phosphoric acid fuel cell · Protonic ceramic fuel cell · Photoelectrochemical cell · Proton exchange membrane fuel cell · Regenerative fuel cell · Reformed methanol fuel cell · Solid oxide electrolyser cell · Solid oxide fuel cell · Unitized regenerative fuel cell · Zinc-air batteryHydrogen Categories:- Fuel cells
- Sustainable technologies
Wikimedia Foundation. 2010.