- Chloramine-T
-
Chloramine-T  N-chloro 4-methylbenzenesulfonamide, sodium saltOther namesN-chloro para-toluenesulfonylamide,
N-chloro 4-methylbenzenesulfonamide, sodium saltOther namesN-chloro para-toluenesulfonylamide,
sodium chloro[(4-methyl phenyl)sulfonyl]azanide, chloramine-TIdentifiers CAS number 127-65-1  , 7080-50-4 (trihydrate)
, 7080-50-4 (trihydrate)PubChem 10198484, 31388 (free acid) ChemSpider 2876055  , 29119 (free acid)
, 29119 (free acid)UNII 4IU6VSV0EI 
KEGG D02445 
ChEBI CHEBI:53782 
ChEMBL CHEMBL1697734 
ATC code D08,QP53AB04 Jmol-3D images Image 1 - [Na+].O=S(=O)([N-]Cl)c1ccc(cc1)C
Properties Molecular formula C7H7ClNO2S·Na (3H2O) Molar mass 227.64 g/mol Appearance white powder Density 1.4 g/cm³ Melting point releases chlorine at 130 °C
solid melts at 167–169 °CSolubility in water 15% in water (25 °C)
7.5% in rectified spirit (20 °C)
insoluble in benzene and ethersHazards R-phrases R22 R31 R34 R42 S-phrases (S1/2) S7 S22 S26 S36/37/39 S45 Main hazards Corrosive  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Tosylchloramide or N-chloro tosylamide, sodium salt, sold as chloramine-T, is a N-chlorinated and N-deprotonated sulfonamide used as a biocide and a mild disinfectant. It is a white powder that gives unstable solutions with water. Trade names of chloramine-T products include Disifin.
Contents
Chemistry
As an N-chloro compound, it contains active (electrophilic) chlorine and can be compared to the O-chlorinated sodium hypochlorite. Chloramine-T is slightly basic (pH typically 8.5). In water, it breaks down to the disinfectant hypochlorite. It can be used as a source of electrophilic chlorine in organic synthesis.
The anionic sulfonamide is stabilized by delocalization of the nitrogen lone pair across the SO2 group, facilitating deprotonation of the parent amine by bases such as hydroxide. It is readily biodegradable by use of ultraviolet (UV) light.
Synthesis
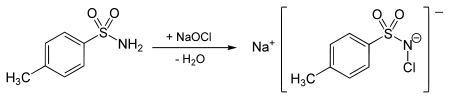
Chloramine-T is prepared from p-toluenesulfonamide and sodium hypochlorite, with the latter being produced in situ from sodium hydroxide and chlorine (Cl2).[1]
Use in iodination and radioiodination
Hypochlorite released from chloramine-T acts as an effective oxidizing agent for iodide to form iodine monochloride (ICl). ICl rapidly undergoes electrophilic substitution predominantly with activated aromatic rings, such as those of the amino acid tyrosine. Thus, chloramine-T is widely used for the incorporation of iodine to peptides and proteins. Chloramine-T together with iodogen or lactoperoxidase is commonly used for labeling peptides and proteins with radioiodine isotopes (123I, 125I or 131I).[2]
Use as a biocide
Chloramine-T is available in tablet or powder form and has to be dissolved before use. It is sprayed on a surface and allowed to stand for at least 15 minutes before being wiped off or allowed to dry. It used in areas such as hospitals, laboratories, nursing homes, funeral homes, medical, dental and veterinary facilities, where control of pathogens is required, for disinfecting surfaces and soaking medical and dental equipment. The substance is also used for parasite control and for drinking water disinfection.
Chloramine-T is as an algicide, bactericide, virucide, fungicide (including spores), germicide. It is also effective against mycobacteria such as tuberculosis, foot-and-mouth disease and avian influenza. The molecular structure of toluenesulfonylamide is similar to para-aminobenzoic acid, an intermediate in bacterial metabolism, which is disrupted by this sulfonamide (in the same way as by a sulfa drug). Therefore, chloramine-T is capable of inhibiting with bacterial growth with two mechanisms, with the phenylsulfonamide moiety and the hypochlorite, which destroys the DNA structure via oxidation and thereby prevents microbes from reproducing and reforming.
Certifications
- EN 1276 Bactericidal
- EN 13713 Bactericidal
- EN 14675 Virucidal
- EN 14476 Virucidal Norovirus
- EN 1650 Fungicidal
- EN 13704 Sporicidal Clostridium difficile
References
- ^ M. Shetty, T. B. Gowda, A Study of Substituent Effect on the Oxidative Strengths of N-Chloroarenesulphonamides: Kinetics of Oxidation of Leucine and Isoleucine in Aqueous Acid Medium, in: Zeitschrift für Naturforschung 2004, 59, 63–72.
- ^ F.Rösch. Radiochemistry and Radiopharmaceutical Chemistry in Life Sciences. Volume 4. Dordrecht/Boston/London: Kluwer Academic Publishers.
External links
- Chemicalland21.com: [1]
- MSDS [2], ICSS [3]
- "Disifin USA". http://www.disifinusa.com. Retrieved 2010-02-09.
Antiseptics and disinfectants (D08) Acridine derivatives Ethacridine lactate • 9-Aminoacridine • EuflavineBiguanides and amidines Phenol and derivatives Nitrofuran derivatives NitrofurazoneIodine products Iodine/octylphenoxypolyglycolether • Povidone-iodine# • DiiodohydroxypropaneQuinoline derivatives Quaternary ammonium compounds Mercurial products Silver compounds Alcohols Other #WHO-EM. ‡Withdrawn from market. Clinical trials: †Phase III. §Never to phase III 
This dermatologic drug article is a stub. You can help Wikipedia by expanding it.
