- Neural development in humans
-
Main article: Neural development
The study of neural development draws on both neuroscience and developmental biology to describe the cellular and molecular mechanisms by which complex nervous systems emerge during embryonic development and throughout life.
Some landmarks of embryonic neural development include the birth and differentiation of neurons from stem cell precursors, the migration of immature neurons from their birthplaces in the embryo to their final positions, outgrowth of axons from neurons and guidance of the motile growth cone through the embryo towards postsynaptic partners, the generation of synapses between these axons and their postsynaptic partners, the neuron pruning that occurs in adolescence, and finally the lifelong changes in synapses which are thought to underlie learning and memory.
Typically, these neurodevelopmental processes can be broadly divided into two classes: activity-independent mechanisms and activity-dependent mechanisms. Activity-independent mechanisms are generally believed to occur as hardwired processes determined by genetic programs played out within individual neurons. These include differentiation, migration and axon guidance to their initial target areas. These processes are thought of as being independent of neural activity and sensory experience. Once axons reach their target areas, activity-dependent mechanisms come into play. Neural activity and sensory experience will mediate formation of new synapses, as well as synaptic plasticity, which will be responsible for refinement of the nascent neural circuits.
Contents
Embryonic stage
Neurulation
Main article: neurulation- See embryogenesis for understanding the animal development up to this stage.
Neurulation is the formation of the neural tube from the ectoderm of the embryo. It follows gastrulation in all vertebrates.
During gastrulation cells migrate to the interior of embryo, forming three germ layers— the endoderm (the deepest layer), mesoderm and ectoderm (the surface layer)—from which all tissues and organs will arise. In a simplified way, it can be said that the ectoderm gives rise to skin and nervous system, the endoderm to the guts and the mesoderm to the rest of the organs.
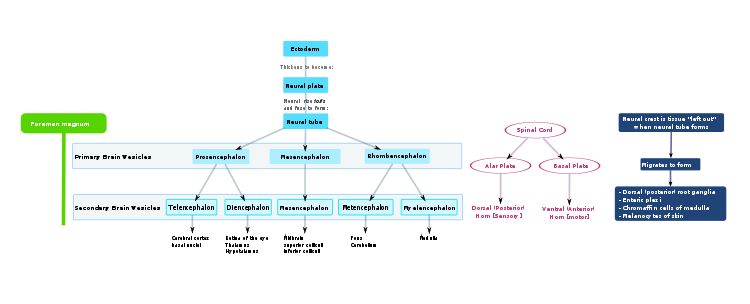
After gastrulation the notochord—a flexible, rod-shaped body that runs along the back of the embryo—has been formed from the mesoderm. During the third week of gestation the notochord sends signals to the overlying ectoderm, inducing it to become neuroectoderm. This results in a strip of neuronal stem cells that runs along the back of the fetus. This strip is called the neural plate, and is the origin of the entire nervous system. The neural plate folds outwards to form the neural groove. Beginning in the future neck region, the neural folds of this groove close to create the neural tube (this form of neurulation is called primary neuralation). The anterior (front) part of the neural tube is called the basal plate; the posterior (rear) part is called the alar plate. The hollow interior is called the neural canal. By the end of the fourth week of gestation, the open ends of the neural tube (the neuropores) close off.[1]
Formation of the spinal cord
The spinal cord forms from the lower part of the neural tube. The wall of the neural tube consists of neuroepithelial cells, which differentiate into neuroblasts, forming the mantle layer (the gray matter). Nerve fibers emerge from these neuroblasts to form the marginal layer (the white matter).
The ventral part of the mantle layer (the basal plates) forms the motor areas of the spinal cord, whilst the dorsal part (the alar plates) forms the sensory areas. Between the basal and alar plates is an intermediate layer that contains neurons of the autonomic nervous system.[2]
Formation of the brain
Late in the fourth week, the superior part of the neural tube flexes at the level of the future midbrain—the mesencephalon. Above the mesencephalon is the prosencephalon (future forebrain) and beneath it is the rhombencephalon (future hindbrain). The optical vesicle (which will eventually become the optic nerve, retina and iris) forms at the basal plate of the prosencephalon.
In the fifth week, the alar plate of the prosencephalon expands to form the cerebral hemispheres (the telencephalon). The basal plate becomes the diencephalon.
The diencephalon, mesencephalon and rhombencephalon constitute the brain stem of the embryo. It continues to flex at the mesencephalon. The rhombencephalon folds posteriorly, which causes its alar plate to flare and form the fourth ventricle of the brain. The pons and the cerebellum form in the upper part of the rhombencephalon, whilst the medulla oblongata forms in the lower part.
Human brain development
See also: Human brain development timelineNeuronal migration
Neuronal migration is the method by which neurons travel from their origin or birth place to their final position in the brain. There are several ways they can do this, e.g. by radial migration or tangential migration.
Radial migration
Neuronal precursor cells proliferate in the ventricular zone of the developing neocortex. The first postmitotic cells to migrate form the preplate which are destined to become Cajal-Retzius cells and subplate neurons. These cells do so by somal translocation. Neurons migrating with this mode of locomotion are bipolar and attach the leading edge of the process to the pia. The soma is then transported to the pial surface by nucleokenisis, a process by which a microtubule "cage" around the nucleus elongates and contracts in association with the centrosome to guide the nucleus to its final destination.[3] Radial fibres (also known as radial glia) can translocate to the cortical plate and differentiate either into astrocytes or neurons.[4][citation needed] Somal translocation can occur at any time during development.[5]
Subsequent waves of neurons split the preplate by migrating along radial glial fibres to form the cortical plate. Each wave of migrating cells travel past their predecessors forming layers in an inside-out manner, meaning that the youngest neurons are the closest to the surface.[6][7] It is estimated that glial guided migration represents 80-90% of migrating neurons.[citation needed]
Tangential migration
Most interneurons migrate tangentially through multiple modes of migration to reach their appropriate location in the cortex. An example of tangential migration is the movement of Cajal-Retzius cells from the cortical hem to the superfitial part of cortical neuroepithelium.
Others
There is also a method of neuronal migration called multipolar migration.[8][9] This is seen in multipolar cells, which are abundantly present in the cortical intermediate zone. They do not resemble the cells migrating by locomotion or somal translocation. Instead these multipolar cells express neuronal markers and extend multiple thin processes in various directions independently of the radial glial fibers.[10]
Neurotrophic factors
Neurotrophic factors are molecules which promote and regulate neuronal survival in the developing nervous system. They are distinguished from ubiquitous metabolites necessary for cellular maintenance and growth by their specificity; each neurotrophic factor promotes the survival of only certain kinds of neurons during a particular stage of their development. In addition, it had been argued that neurotropihic factors are involved in many other aspects of neuronal development ranging from axonal guidance to regulation of neurotransmitter synthesis. [11]
Neural development in the adult nervous system
Main article: NeuroregenerationNeural development in the adult nervous system includes mechanisms such as remyelination, generation of new neurons, glia, axons, myelin or synapses. Neuroregeneration differs between the peripheral nervous system (PNS) and the central nervous system (CNS) by the functional mechanisms and especially, the extent and speed.
See also
- Time lapse seqeunces of radial migration (also known as glial guidance) and somal translocation.[5]
- Axon guidance
- Neural Darwinism
- Pre- and perinatal psychology
- Neural development
References
- ^ Estomih Mtui; Gregory Gruener (2006). Clinical Neuroanatomy and Neuroscience. Philadelphia: Saunders. pp. 1. ISBN 1-4160-3445-5.
- ^ Atlas of Human Embryology, Chronolab. Last accessed on Oct 30, 2007.
- ^ Samuels B, Tsai L (2004). "Nucleokinesis illuminated". Nat Neurosci 7 (11): 1169–70. doi:10.1038/nn1104-1169. PMID 15508010. http://www.nature.com/neuro/journal/v7/n11/full/nn1104-1169.html.
- ^ Campbell K, Götz M (May 2002). "Radial glia: multi-purpose cells for vertebrate brain development". Trends Neurosci. 25 (5): 235–8. doi:10.1016/S0166-2236(02)02156-2. PMID 11972958. http://linkinghub.elsevier.com/retrieve/pii/S0166223602021562.
- ^ a b Nadarajah B, Brunstrom J, Grutzendler J, Wong R, Pearlman A (2001). "Two modes of radial migration in early development of the cerebral cortex". Nat Neurosci 4 (2): 143–50. doi:10.1038/83967. PMID 11175874. http://www.nature.com/neuro/journal/v4/n2/full/nn0201_143.html.
- ^ Nadarajah B, Parnavelas J (2002). "Modes of neuronal migration in the developing cerebral cortex". Nat Rev Neurosci 3 (6): 423–32. doi:10.1038/nrn845. PMID 12042877.
- ^ Rakic P (1972). "Mode of cell migration to the superficial layers of fetal monkey neocortex". J Comp Neurol 145 (1): 61–83. doi:10.1002/cne.901450105. PMID 4624784.
- ^ Tabata H, Nakajima K (5 November 2003). "Multipolar migration: the third mode of radial neuronal migration in the developing cerebral cortex". J Neurosci 23 (31): 9996–10001. PMID 14602813. http://www.jneurosci.org/cgi/content/full/23/31/9996.
- ^ Nadarajah B, Alifragis P, Wong R, Parnavelas J (2003). "Neuronal migration in the developing cerebral cortex: observations based on real-time imaging". Cereb Cortex 13 (6): 607–11. doi:10.1093/cercor/13.6.607. PMID 12764035. http://cercor.oxfordjournals.org/cgi/content/full/13/6/607.
- ^ Tabata H, Nakajima K (5 November 2003). "Multipolar migration: the third mode of radial neuronal migration in the developing cerebral cortex". J Neurosci. 23 (31): 9996–10001. PMID 14602813. http://www.jneurosci.org/cgi/pmidlookup?view=long&pmid=14602813.
- ^ Alan M. Davies (1 may 1988)"Trends In Genetics", Volume 4-Issue 5; Department of Anatomy, St George's Hospital Medical School, Cranmer Terrace, Tooting, London SW17 0RE, UK
Prenatal development/Mammalian development of nervous system (GA 9.733 and GA 10.1002, TE E5.13-16) Neurogenesis Cranial neural crest (Cardiac neural crest complex) · Truncal neural crestRostral neuropore
Cephalic flexure · Pontine flexure
Alar plate (sensory) · Basal plate (motor)
Germinal matrixEye development Auditory development M: EYE
anat(g/a/p)/phys/devp/prot
noco/cong/tumr, epon
proc, drug(S1A/1E/1F/1L)
M: EAR
anat(e/p)/phys/devp
noco/cong, epon
proc, drug(S2)
Categories:- Developmental biology
- Embryology of nervous system
- Developmental neuroscience
Wikimedia Foundation. 2010.