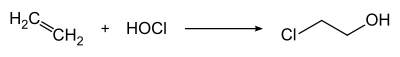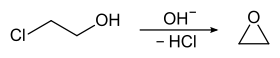- 2-Chloroethanol
-
2-Chloroethanol 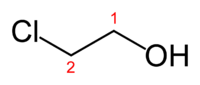
 2-ChloroethanolOther names2-chloroethyl alcohol, ethylene chlorohydrin, glycol chlorohydrin, 2-chloro-1-ethanol, 2-monochloroethanol, 2-hydroxyethyl chloride, β-chloroethanol, β-hydroxyethyl chloride, chloroethanol, δ-chloroethanol, ethylchlorhydrin, ethylene chlorohydrin, glycol monochlorohydrin
2-ChloroethanolOther names2-chloroethyl alcohol, ethylene chlorohydrin, glycol chlorohydrin, 2-chloro-1-ethanol, 2-monochloroethanol, 2-hydroxyethyl chloride, β-chloroethanol, β-hydroxyethyl chloride, chloroethanol, δ-chloroethanol, ethylchlorhydrin, ethylene chlorohydrin, glycol monochlorohydrinIdentifiers CAS number 107-07-3 
ChemSpider 21106015 
UNII 753N66IHAN 
KEGG C06753 
ChEBI CHEBI:28200 
ChEMBL CHEMBL191244 
Jmol-3D images Image 1 - ClCCO
Properties Molecular formula C2H5ClO Molar mass 80.52 g/mol Density 1.197 g/cm³ Melting point -67 °C
Boiling point 128-130 °C
Related compounds Related compounds 2-Fluoroethanol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references 2-Chloroethanol is an organochlorine compound with the formula HOCH2CH2Cl. This colorless liquid has a pleasant ether-like odor. It is miscible with water. The molecule is bifunctional, consisting of both an alkyl chloride and an alcohol functional groups.
Contents
Synthesis and applications
2-Chloroethanol is produced by treating ethylene with hypochlorous acid:
2-Chloroethanol was once produced on a large scale as a precursor to ethylene oxide:
This application has been supplanted by the greener direct oxidation of ethylene. Otherwise chloroethanol is used in a number of specialized applications.[1] Several dyes are prepared by the alkylation of aniline derivatives with chloroethanol.[2] It is a building block in the production of pharmaceuticals, biocides and plasticizers. It is also used for manufacture of thiodiglycol. It is a solvent for cellulose acetate and ethyl cellulose, textile printing dyes, in dewaxing, refining of rosin, extraction of pine lignin, and the cleaning of machines.
Environmental aspects
Chloroethanol is a metabolite in the degradation of 1,2-dichloroethane. The alcohol is then further oxidized via chloroacetaldehyde to chloroacetate. This metabolic pathway is topical since billions of kilograms of 1,2-dichloroethane are processed annually as a precursor to vinyl chloride.[3]
Safety
2-Chloroethanol is toxic with an LD50 of 89 mg/kg in rats. Like most organochlorine compounds, chloroethanol combusts to yield hydrogen chloride and phosgene.
References
- ^ Gordon Y. T. Liu, W. Frank Richey, Joanne E. Betso “Chlorohydrins“ in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_565
- ^ Roderich Raue and John F. Corbett “Nitro and Nitroso Dyes“ in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_383
- ^ 1. Janssen, D. B.; van der Ploeg, J. R. and Pries, F., "Genetics and Biochemistry of 1,2-Dichloroethane Degradation", Biodegradation, 1994, volume 5, pp. 249-57.doi:10.1007/BF00696463
Categories:- Alcohols
- Organochlorides
Wikimedia Foundation. 2010.