- Organotitanium compound
-
 Organotitanium compounds
Organotitanium compounds
Organotitanium compounds in organometallic chemistry contain carbon to titanium chemical bonds. Organotitanium chemistry is the science of organotitanium compounds describing their physical properties, synthesis and reactions. They are reagents in organic chemistry and are involved in major industrial processes.[1][2]
Contents
Brief history
Although the first attempt at an organotitanium compound dates back to 1861, it took until 1953 for the first such compound to arrive. In that year titanium phenyl-tri(isopropoxide) was prepared by Herman and Nelson from titanium isopropoxide, phenyllithium and titanium(IV) chloride. Titanocene dichloride was invented in 1954, and the first methyltitanium compounds were introduced in 1959. Ziegler-Natta catalysts utilizing titanium-based catalysts soon followed as a major commercial application for which the Nobel Prize in Chemistry of 1963 was awarded.
Properties
The titanium electron configuration ([Ar]3d24s2) resembles that of carbon and like carbon the +4 oxidation state dominates and like carbon compounds, those of titanium have a tetrahedral molecular geometry. Thus, the boiling points of TiCl4 and CCl4 are very similar. Titanium is however a much larger element than carbon, reflected by the Ti-C bond lengthes being about 30% longer, e.g. 210 pm in tetrabenzyltitanium vs a typical C-C bond of 155 pm. Simple tetraalkyltitanium compounds however are not typically stable, owing to the large size of titanium and the electron-deficient nature of its tetrahdral complexes. More abundant and more useful than the simple tetraalkyl compounds are organic derivatives with alkoxide and cyclopentadienyl coligands. Titanium is capable of forming complexes with high coordination numbers.
In terms of oxidation states, most organotitanium chemistry, in solution at least, focuses on derivatives of Ti(IV). Ti(II) compounds are rarer, examples being titanocene dicarbonyl and Ti(CH3)2(dmpe)2. [Ti(CO)6]2- is formally a complex of Ti(-II).[3] Although Ti(III) is involved in Ziegler-Natta catalysis, the organic derivatives of Ti(III) are not common.
Due to the low electronegativity of titanium, Ti-C bonds are polarized toward carbon. Consequently, alkyl ligands in many titanium compounds are nucleophilic. Titanium is characteristically oxophilic, which presents challenges to handling these compounds, which require air-free techniques. On the other hand, high oxophilicity means that titanium alklyls are effective for abstracting or exchanging organyl ligands for oxo groups, as discussed below.
Chemistry
Organotitanium compounds are important reagents in organic chemistry.[4] Some reagents include the following.
- The Ziegler-Natta catalyst (1954) is obtained from titanium(III) chloride and diethylaluminium chloride and important in ethylene polymerization.
- Methyl titanium trichloride CH3TiCl3 (1959) is a nonbasic nucleophilic reagent. It can be prepared by reacting titanium(IV) chloride with dimethylzinc in dichloromethane at -78 °C. It is used in nucleophilic addition of methyl groups to carbonyl compounds and in SN1 methylation of alkyl halides. Methyltriisopropoxytitanium is a related reagent prepared in situ from titanium isopropoxide, titanium(IV) chloride and methyllithium[5]
- The Kulinkovich reaction is a cyclopropanation method starting from a Grignard reagent and an ester. The first step is transmetallation forming a dialkyltitanium intermediate.
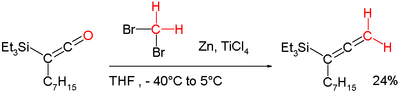
- Lombardo's reagent is a carbenoid methylenation reagent (see Tebbe reagent below).[6], which is an aged version of the Dibromomethane-Zinc-Titanium(IV) Chloride reagent[7] can for example be applied in a conversion of a ketene into an allene:[8][4]
Titanocene derivatives
A particularly rich area of organotitanium chemistry are derivatives of titanocene dichloride. Early work on "titanocene" itself eventually revealed that this species was a fulvalene complex, and this discovery led to many innovations on cyclopentadienyl complexes of titanium.
Tebbe's reagent (1978) is prepared from titanocene dichloride and trimethylaluminium. It is used as a methylenation agent for carbonyl compounds (converstion of R2C=O to R2C=CH2). It is an alternative for Wittig reagents when the carbonyl group is sterically challenged or when it easily forms the enol.[4] Tebbe's reagent adds simple alkenes to give titanocyclobutanes, which can be regarded as stable olefin metathesis intermediates. These compounds are reagents in itself such as 1,1-bis(cyclopentadienyl)-3,3-dimethyltitanocyclobutane, the adduct of Tebbe's reagent with isobutene catalysed with 4-dimethylaminopyridine.[9]
The Petasis reagent or dimethyl titanocene (1990) is prepared from titanocene dichloride and methyllithium in diethyl ether. Compared to Tebbe's reagent it is easier to prepare and easier to handle. It is also a methylenation reagent.[9]
See also
CH He CLi CBe CB CC CN CO CF Ne CNa CMg CAl CSi CP CS CCl CAr CK CCa CSc CTi CV CCr CMn CFe CCo CNi CCu CZn CGa CGe CAs CSe CBr CKr CRb CSr CY CZr CNb CMo CTc CRu CRh CPd CAg CCd CIn CSn CSb CTe CI CXe CCs CBa CHf CTa CW CRe COs CIr CPt CAu CHg CTl CPb CBi CPo CAt Rn Fr Ra Rf Db Sg Bh Hs Mt Ds Rg Cn Uut Uuq Uup Uuh Uus Uuo ↓ CLa CCe CPr CNd CPm CSm CEu CGd CTb CDy CHo CEr CTm CYb CLu Ac Th Pa CU Np Pu Am Cm Bk Cf Es Fm Md No Lr Chemical bonds to carbon Core organic chemistry Many uses in chemistry Academic research, but no widespread use Bond unknown / not assessed References
- ^ "Encyclopedia of Reagents for Organic Synthesis", L.A. Paquette, Ed.: J. Wiley and Sons: Sussex, England, 1996
- ^ "Organotitanium Reagents in Organic Synthesis (Reactivity and Structure Concepts in Organic Chemistry, Vol 24)" Manfred T. Reetz 1986 ISBN 0-387-15784-0
- ^ Elschenbroich, C. ”Organometallics” (2006) Wiley-VCH: Weinheim. ISBN 978-3-29390-6
- ^ a b c Hartwig, J. F. Organotransition Metal Chemistry, from Bonding to Catalysis; University Science Books: New York, 2010. ISBN 189138953X
- ^ Organic Syntheses, Coll. Vol. 8, p.495 (1993); Vol. 67, p.180 (1989) Link.
- ^ Organic Syntheses, Coll. Vol. 8, p.386 (1993); Vol. 65, p.81 (1987) Link.
- ^ Takai, K.; Hotta, Y.; Oshima, K.; Nozaki, H. Tetrahedron Lett. 1978, 2417-2420.
- ^ "Synthesis of highly substituted allenylsilanes by alkylidenation of silylketenes" Stephen P Marsden and Pascal C Ducept Beilstein Journal of Organic Chemistry 2005, 1:5 doi:10.1186/1860-5397-1-5
- ^ a b "Titanium carbenoid reagents for converting carbonyl groups into alkenes" Hartley, R. C.; Li, J.; Main, C. A.; McKiernan, G. J. Tetrahedron 2007, 63, 4825-4864 (Review) doi:10.1016/j.tet.2007.03.015.
Categories:- Organotitanium compounds
Wikimedia Foundation. 2010.